Audience:
- Transplant program directors, administrators, data coordinators, clinical coordinators, compliance and quality managers, and clinical support staff
- All member representatives and alternate representatives
Implementation date: March 17, 2020
At-a-glance
Effective immediately, new policy language is in effect regarding data submission requirements for transplant candidates in cases where data collection is affected by the COVID-19 outbreak. The OPTN Executive Committee approved this action on March 17, 2020, as an emergency policy per the OPTN bylaws; it will review the policy at least every three months to assess whether to continue it.* It only applies to circumstances directly related to the COVID-19 emergency. Details of the policy are contained in the policy notice on the OPTN website.
In separate action, a new candidate temporarily inactive reason code, “COVID-19 precaution,” has been added to WaitlistSM to address instances where a transplant program opts to temporarily defer organ offers for a transplant candidate due to issues relating to the COVID-19 outbreak. This is an operational action separate from an OPTN policy or bylaw change.
The Executive Committee of the OPTN, at a virtual meeting held July 30, extended the expiration date for a set of operational actions to help members address and document COVID-19 issues affecting organ donation and transplantation. The measures will stay in effect, subject to ongoing review of their applicability and effectiveness.
What you need to do
In all instances, continue to exercise your medical judgment in providing care for your transplant candidates. If issues relating to COVID-19 alter your usual decisions or actions, please document this in the candidate’s medical record.
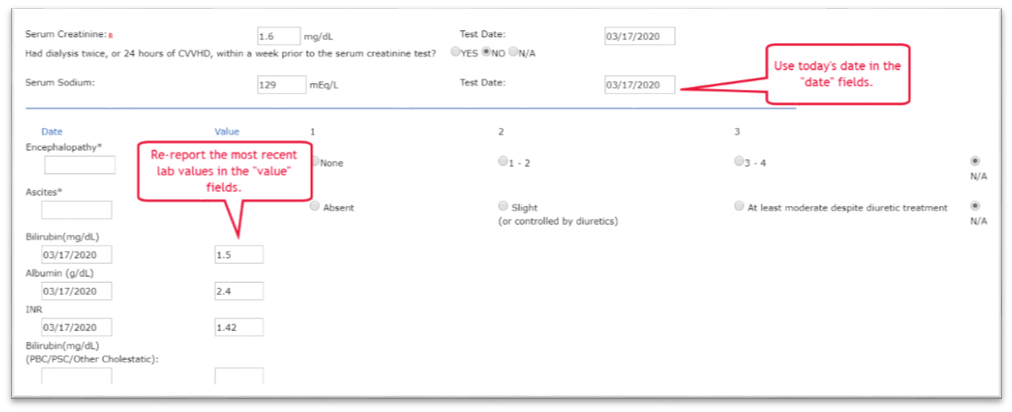
For maintaining data to support a transplant candidate’s listing status or waiting time, please use the most updated clinical data you have available and make all reasonable efforts to meet OPTN requirements. If your program is unable to collect updated data due to issues related to COVID-19, or if in your medical judgment you opt not to collect updated data because of COVID-19 considerations, you may report the most recent clinical data values you previously submitted. In this instance, please report today’s date (the date you are submitting the data) as the actual date of the test.
Illustrated example
Submission of new MELD lab values for a liver candidate to prevent downgrade due to an inability to obtain new labs as a result of COVID-19:
An example of acceptable documentation would be a note in the candidate’s medical record such as, “3/20/2020 – updated candidate record in Waitlist. Due to COVID-19 emergency actions, candidate’s previously reported clinical data was reported with today’s date.”
The “COVID-19 precaution” temporarily inactive reason code is available for any individual candidate that, using your medical judgment, may not benefit from receiving organ offers at this time. Again, please document supporting information in the candidate’s medical record.
Questions?
If you have questions relating to implementation, contact UNOS Customer Service at 800-978-4334.
For policy-related questions, send an e-mail to [email protected].